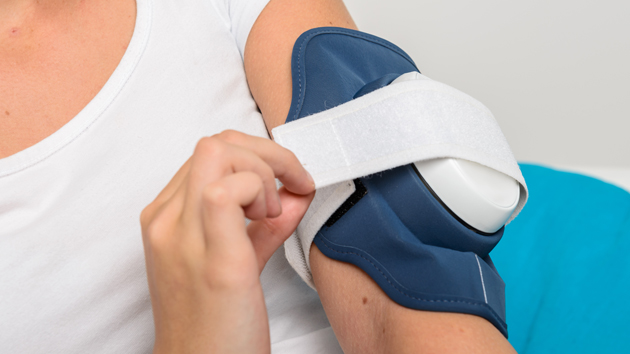
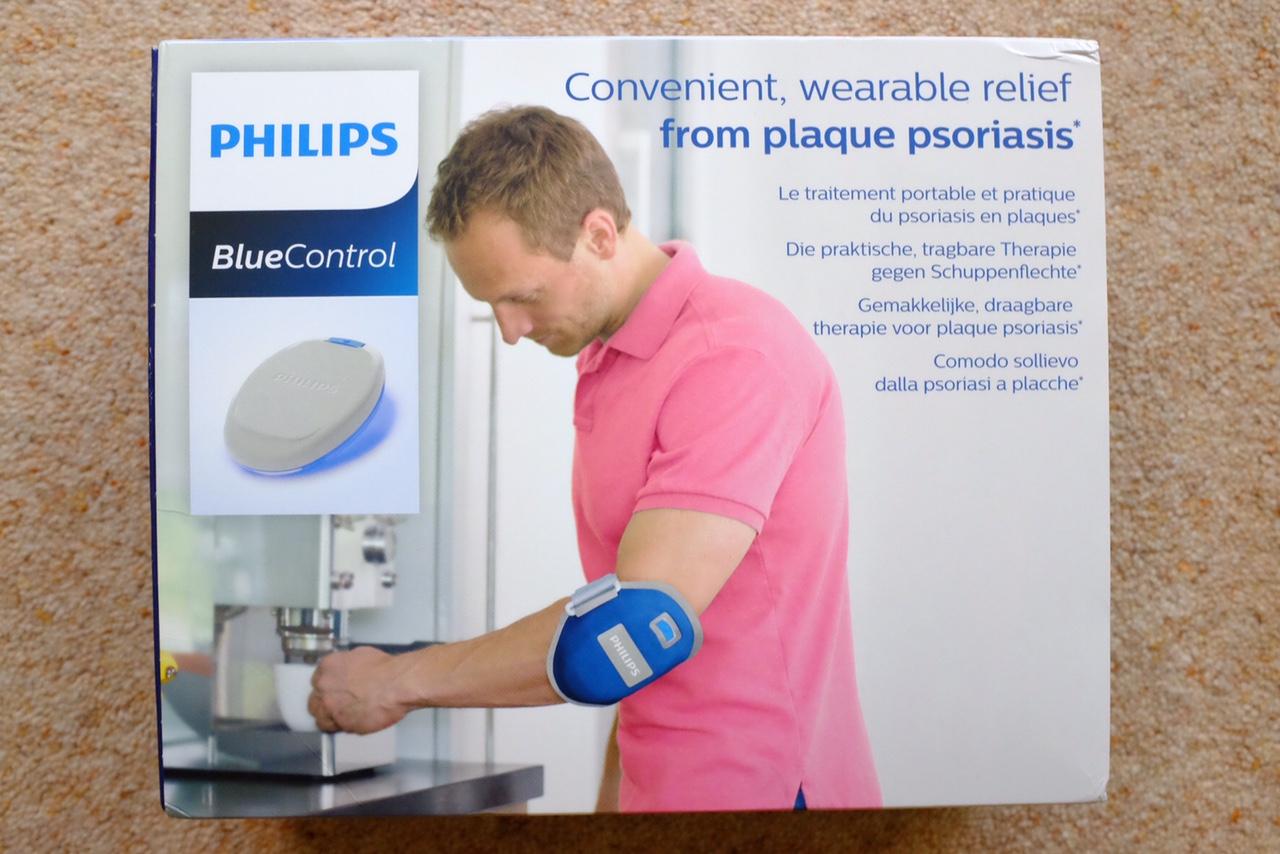
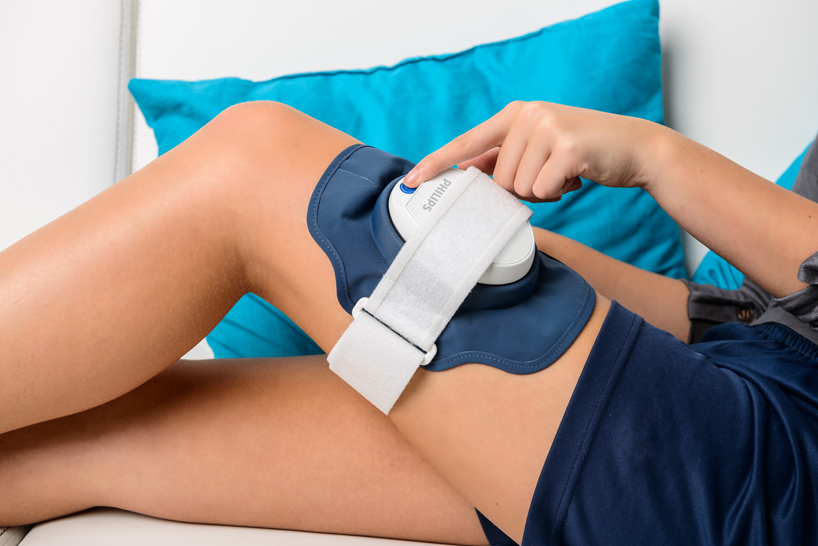
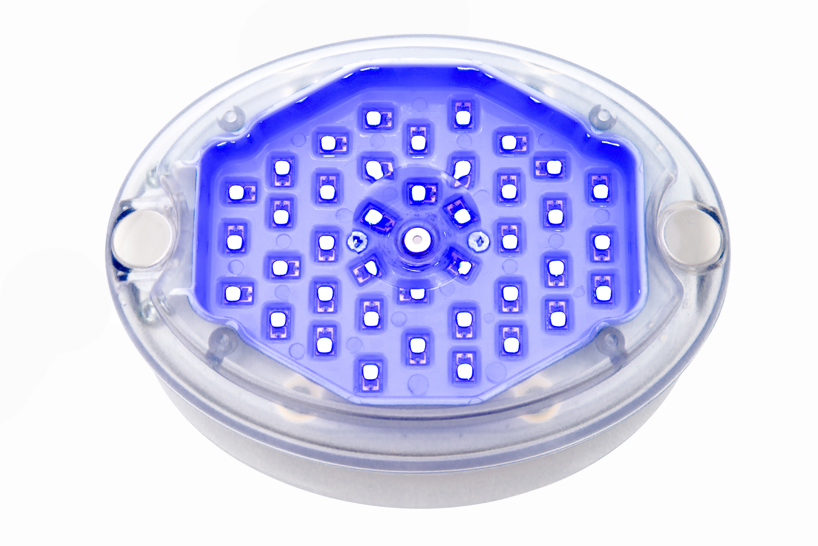
PSK2103/10 Philips BlueControl Advance PSK2103 15 minutes treatment per plaque Wearable blue light therapy Clinically proven efficacy Without chemicals or cortisone - Philips Support

Philips receives U.S. FDA 510(k) clearance to market its Philips BlueControl, a wearable light therapy device to treat mild psoriasis at home - News center | Philips